This page is not an official page of the app or its developer, but an independent editorial publication created for informational and commentary purposes. Unless expressly stated otherwise, neither the app nor its developer is affiliated with, endorsed by, sponsored by, authorized by, or otherwise officially connected with MWM, Apple, Google Play, the app publisher, or the app's developer, and nothing on this page implies that the app was developed using MWM's services. Any trademarks, logos, screenshots, and other content remain the property of their respective owners.
Downloads
0User Rating
Total Ratings
0Publisher
Category
MedicalLocales
68Latest Version
2025.6.1Size
58.3 MBFirst Released
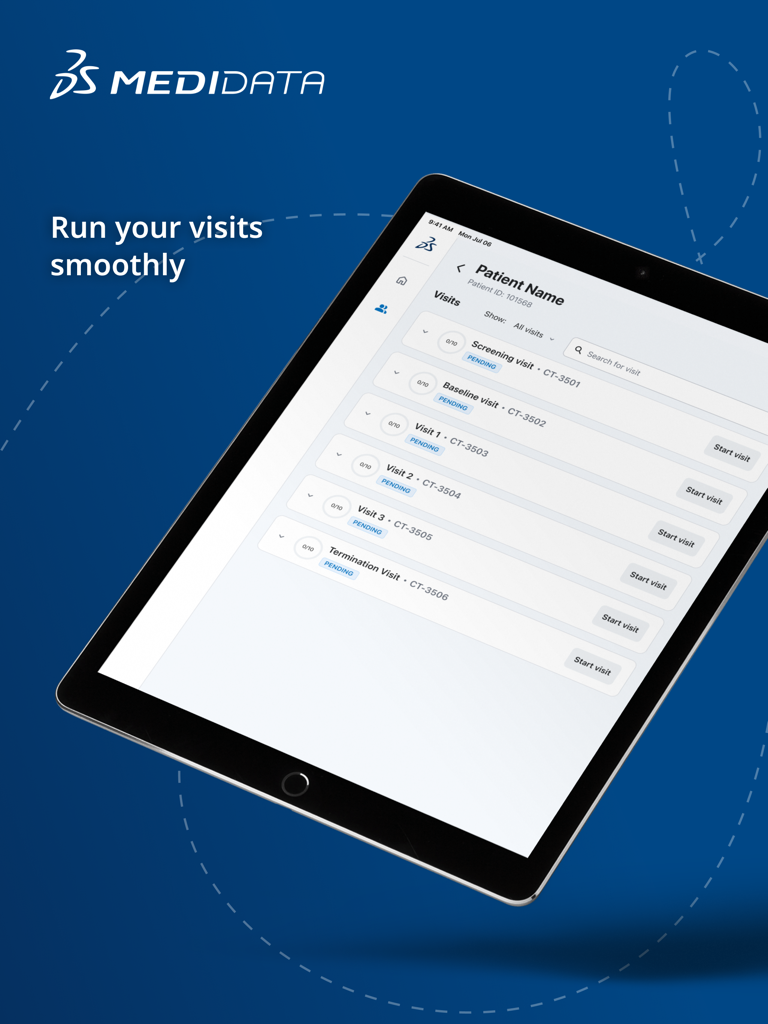
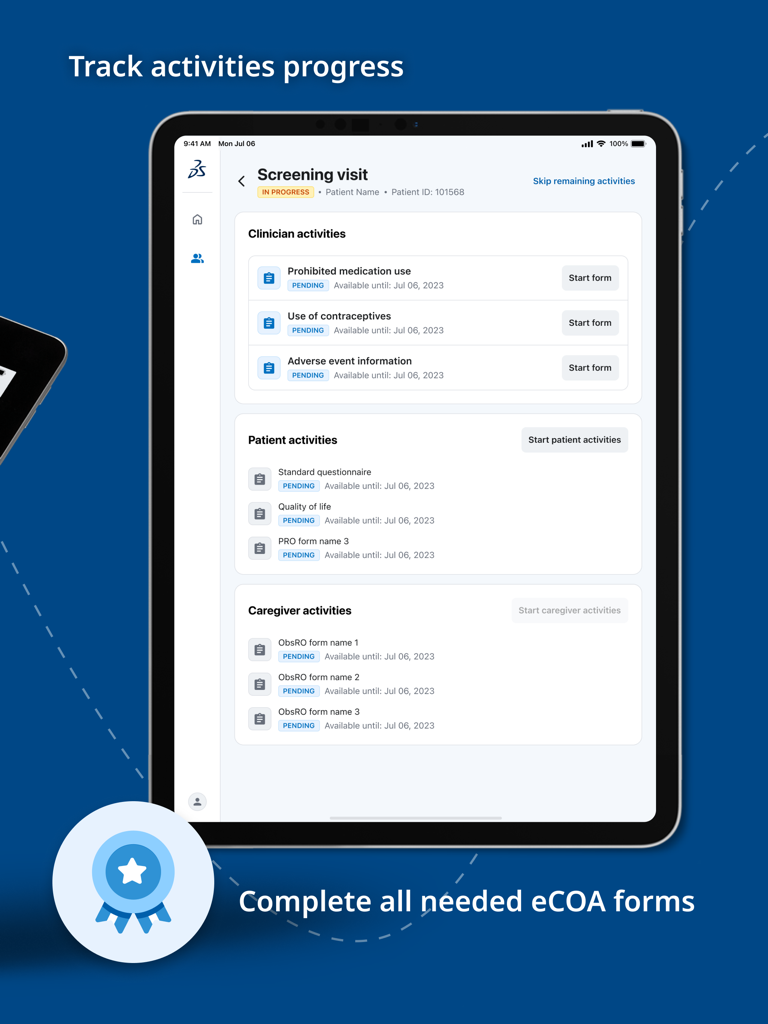
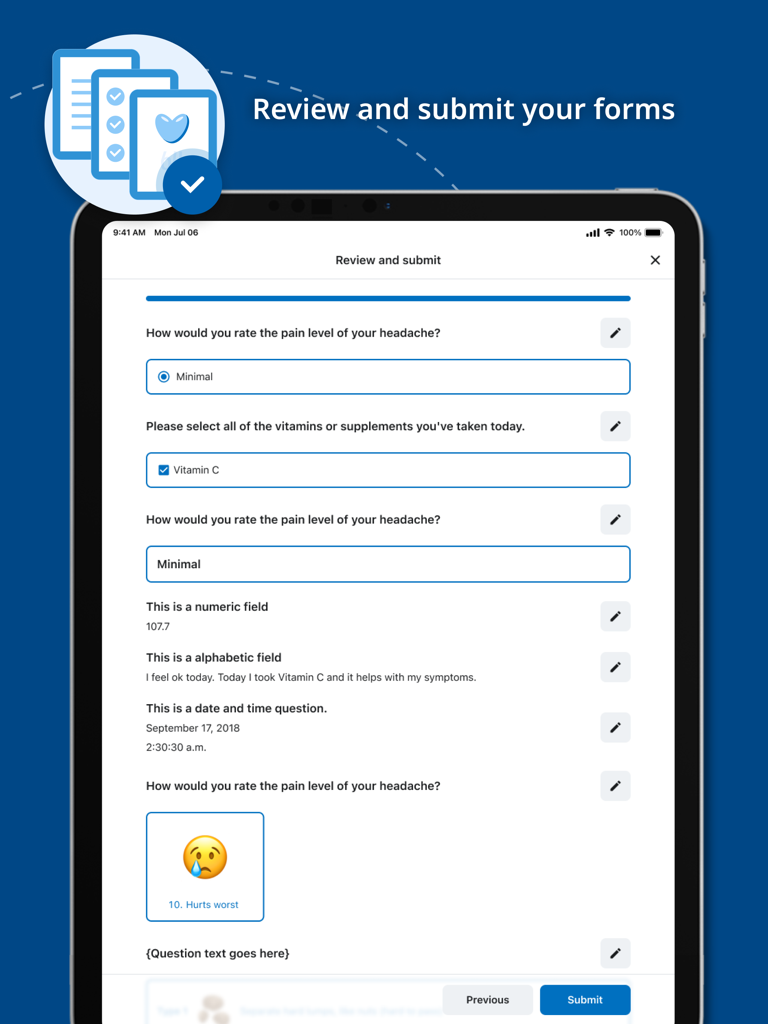
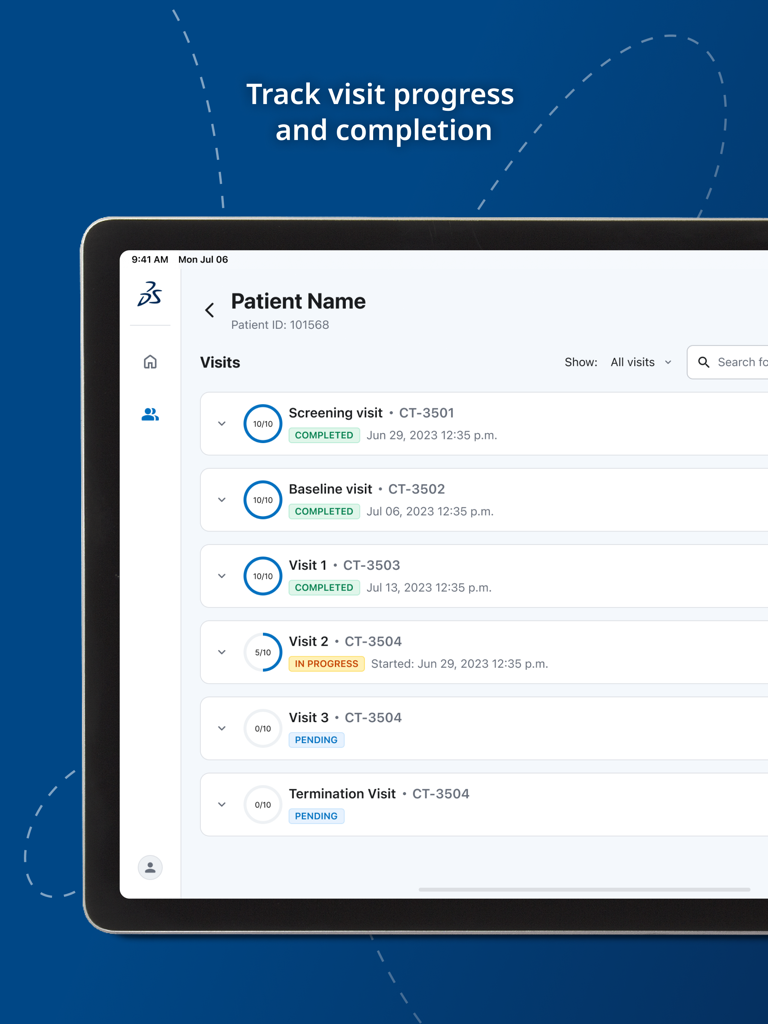
Jul 10, 2025The Future of Clinical Data Collection
Bridge the gap between researchers and patients with a unified platform for ePRO and ClinRO, designed for data integrity and real-time insights.
Seamless Workflow Integration
Directly sync patient data with Medidata Rave to eliminate manual entry errors and accelerate your study timelines.
Hardware Flexibility
Eliminate proprietary device costs by leveraging site tablets or patient devices for a modern, cost-effective trial experience.
More Like This
Top-ranked apps in the same category

MyChart
Epic Systems Corporation

国家医保服务平台
National Healthcare Security Administration

Pregnancy + | Tracker App
Philips Digital UK Limited

CVS Health
CVS Pharmacy

Doctolib - Your health partner
Doctolib

Meu SUS Digital
Servicos e Informacoes do Brasil

Mobile JKN
BPJS Kesehatan

UnitedHealthcare
UNITED HEALTHCARE SERVICES, INC.

江苏医保云
Jiangsu Healthcare Security Bureau
This page is not an official page of the app or its developer, but an independent editorial publication created for informational and commentary purposes. Unless expressly stated otherwise, neither the app nor its developer is affiliated with, endorsed by, sponsored by, authorized by, or otherwise officially connected with MWM, Apple, Google Play, the app publisher, or the app's developer, and nothing on this page implies that the app was developed using MWM's services. Any trademarks, logos, screenshots, and other content remain the property of their respective owners.